When people think about alternative fuel vehicles, Daihatsu is not a name that usually comes to mind. Daihatsu is one of the smaller Japanese car-makers and they sold a couple of models in the US market from 1988-92. More recently, Toyota bought a majority stake in the company and Daihatsu actually assembles most of the models that are sold as Scions in the US market.
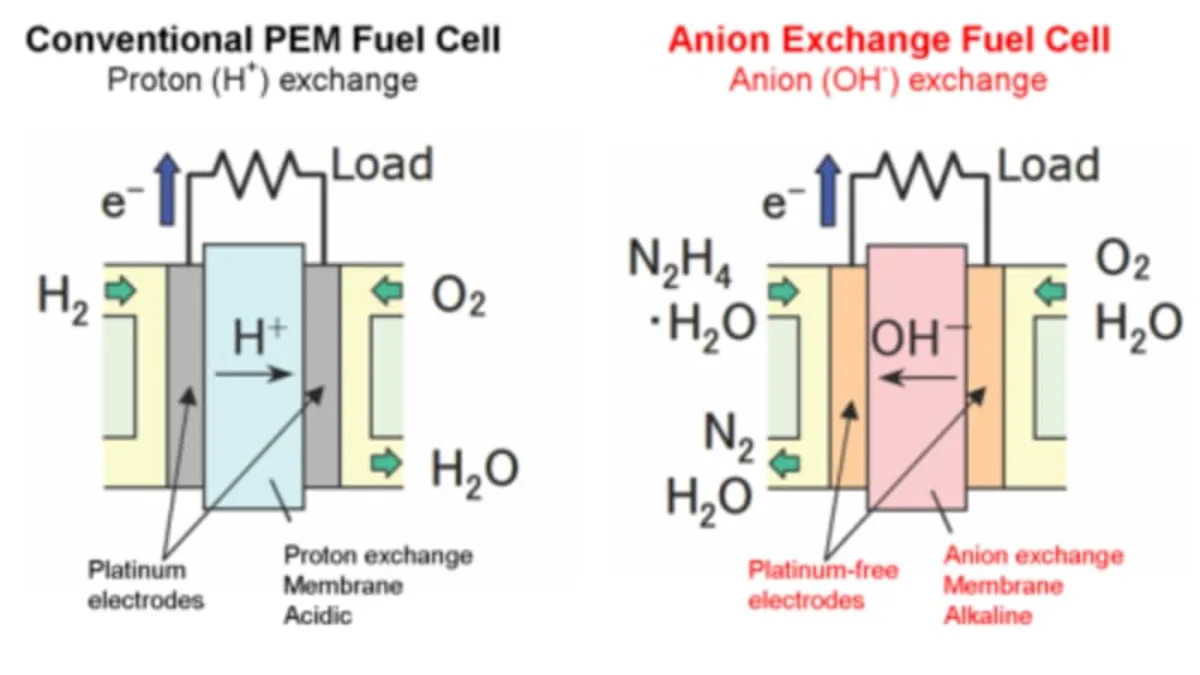
Daihatsu has just announced the development of a new fuel cell that addresses two of the biggest issues with the technology. Unlike other cells, Daihatsus design uses no platinum or other precious metals as a catalyst. Current cells use an acidic electrolyte membrane that protons from the hydrogen pass through. The platinum is used as a catalyst because of its corrosion resistance in the presence of the acidic membrane.
Daihatsu has switched to an alkaline anion exchange membrane. That allows them to use materials such as nickel or cobalt as a catalyst. Instead of hydrogen, Daihatsu is using liquid hydrazine-hydrate as fuel. The fuel is synthetic and more easily handled and transported than hydrogen. They are claiming that they get similar power density of about 0.5 W/cm2 compared to hydrogen fuel cells. This may be an interesting approach to follow, especially if Toyota decides to pursue it.
[Source: Daihatsu]
Daihatsu has just announced the development of a new fuel cell that addresses two of the biggest issues with the technology. Unlike other cells, Daihatsus design uses no platinum or other precious metals as a catalyst. Current cells use an acidic electrolyte membrane that protons from the hydrogen pass through. The platinum is used as a catalyst because of its corrosion resistance in the presence of the acidic membrane.
Daihatsu has switched to an alkaline anion exchange membrane. That allows them to use materials such as nickel or cobalt as a catalyst. Instead of hydrogen, Daihatsu is using liquid hydrazine-hydrate as fuel. The fuel is synthetic and more easily handled and transported than hydrogen. They are claiming that they get similar power density of about 0.5 W/cm2 compared to hydrogen fuel cells. This may be an interesting approach to follow, especially if Toyota decides to pursue it.
[Source: Daihatsu]

Sign in to post
Please sign in to leave a comment.
Continue